Hi all! Some of you may have read my post on a simple chemical method for inducing metamorphosis in a broad diversity of jelly species. In that post I outlines what I did in general terms, but here I’d like to get more specific. Hopefully this will be helpful to fellow jelly wranglers, and of course please feel free to reach out with additional questions either by email (in the “About” section), or in the comment section below.
When is the best time to use this method?
If your polyps have been sitting there doing nothing (don’t we all have some of those…), if you need strobila/ephyrae in a very specific time frame (this is very true for science), or if you have some polyps of uncertain identity, and want to grow up ephyra to figure out who they are, then this method is great for you! That being said, sick or underfed polyps don’t produce the best results. I’ve tried, and with long-starved polyps, or polyps in poor or inappropriate conditions (ie too hot or cold, salinity wrong, etc), the strobilae have a much higher rate of abnormalities (such as spiral strobilation) and the ephyrae will be slow and often missing arms or with general poor symmetry. Much better to prep polyps by feeding them well until they’re fat, and keeping them in clean water at a temperature and salinity they like (no need to match “strobilation conditions,” though it doesn’t hurt).
Which chemical should I use? Are they dangerous?
You have two chemicals to choose from: indomethacin and 5-methoxy-2-methyl-indole (5M2M). The chemical that you choose will depend on which species you want to work with. For everything except box jellies, both indomethacin and 5M2M will work. If you check out Table 1 of my paper, you’ll see that 5M2M always works faster than indomethacin, and for species that take a long time in indomethacin (like Cassiopea), that’s something to consider. Beyond timing, here are some comparisons between the two compounds:
Safety:
Indomethacin is a very well-studied drug that people actually take. This means we know a LOT about its safety and health impacts. Ironically, because of that, it is also listed as a potentially dangerous compound, but the main reason for that is because if you take too much of the drug it causes major bleeding in the digestive tract. However, because it’s so well know, if something were to happen (an intern accidentally eats your whole supply?), doctors will have a good idea of how to treat it. Being well known also means there are great instructions out there on how to handle it safely in all sorts of different situations.
In contract, let’s talk about, 5M2M: while there are no indications that it’s unsafe, it also hasn’t been well studied. No one swallows pills of it regularly, it’s not being researched in a bunch of labs, hundreds of rats and mice and fish haven’t been exposed to it for testing…This means it may be easier to obtain and the guidelines for disposal aren’t as strict, but for me, I like to take extra care, and treat it as if it could potentially be dangerous. This means full lab gear, fume hoods, and careful disposal in a hazardous waste stream.
Efficacy:
Indomethacin isn’t as powerful as 5M2M at inducing metamorphosis. On the surface you might think this is a bad thing, but it can also be a good thing. With both compounds, I get nervous about contamination, and use them in a separate room away from my primary polyp cultures, but 5M2M is really powerful, and I am extra careful with that compound. Even a few drops of 5M2M working solution on the tip of a carelessly-placed pipette could be enough to contaminate cultures and trigger mass strobilation. If I need ephyrae FAST, 5M2M is the way to go, it works twice as fast as indomethacin on every species I tested. But with a little more planning (a couple weeks) indomethacin is fine. In summary: 5M2M is great for quick turn around time, but indomethacin is less risky in terms of contamination. With both, best to be extra careful.
Polyps will strobilate themselves into oblivious if you’re not careful
For both drugs, if you leave polyps soaking they will strobilate forever (pictured below), but I’ve had some pretty good success with recovering polyps. I haven’t done an exhaustive test of recovery methods, but for indomethacin, rinsing Aurelia strobilae really well 3 times in ~50ml of clean water (per 20 polyps) is enough to recover primary polyps.
For 5M2M, 3 hours of exposure is sufficient to induce Southern Japan Aurelia polyps, and you need to wash your animals really well in order for the polyps to re-form. This is how I wash in the lab: I remove each polyp from the soaking solution with a pipette (max 20 polyps), transferring as little solution as possible, and place the polyps in a new 50ml tube of clean water. Then I let them sit for 20 minutes, sometimes gently rolling the tube to encourage water flow. Then, using a new clean pipette, I again move the polyps to a new tube of clean water, etc etc at least five times. Even then, sometimes Chrysaora chesapeakei will continue to strobilate and require even more washes in the following days. It’s important to carefully monitor newly washed strobila under magnification to ensure primary tentacles have reformed and no ephyra furrows are forming below those new primary tentacles. The power of 5M2M is really interesting biologically, but it can be somewhat wash-intensive.
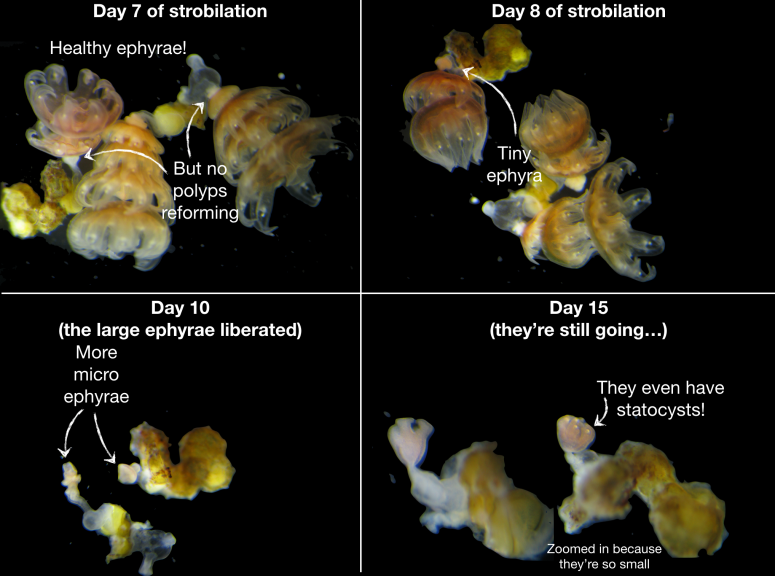
Here’s an example of forever-strobilae in indomethacin. Pacific stinging nettle polyps just going going going until they’re gone:
Polyps and ephyrae in quarantine
For me, it’s easy to keep my animals separated, because I don’t have a shared sump system. I realize some aquariums have a more complex and high tech arrangement. Because my strobila needs are relatively low, I use 50ml falcon tubes, with about 20 polyps per tube. Each tube has 30ml induction solution, I close all the tubes tightly, and let them site tilted on their sides in a low traffic place while induction takes place. After induction I dispose of the tubes and liquid in a hazardous waste container.
For larger volumes, my advice would be to keep polyps that are being induced in a separate area, with separate supplies (pipettes, bubblers, etc), in containers that can be disposed of. Once strobilae have been washed, and polyps re-form at the base, I’d continue to keep them separated until a few days after the final ephyrae has been liberated, just to make sure the new polyps are happy and healthy and not going to start strobing again. If they’re healthy and feeding for about a week with no signs of excess strobing, that’s the best indication they’re clean of the compounds and ready to rejoin the group. If they start strobing, especially if the ephyrae are small (like the pic above) repeat the wash and wait steps.
I wish I had good advice for ephyrae, but the drug doesn’t seem to harm them in any way, and so I don’t worry as much about washing them. But I have had good success using a fine mesh to separate ephyrae from the water. If I had to wash ephyrae, I’d probably start with a similar approach as above (lots of water, lots of washes). I’d be curious to hear what other folks recommend.
But are the jellies healthy?
I’ve grown ephyrae produced with this method up to reproductive maturity for both bay nettles (Chrysaora chesapeakei, formerly known as Chrysaora quinquecirrha) and Southern Japan moons. In fact, here’s a picture of one of my C. chesapeakei, that was strobed with indomethacin (photo by my friend Stefan Siebert):

I haven’t tried growing other species to maturity (I don’t have much space to grow adults), but I would expect them to do just as well.
What species does it work on?
In addition to my paper, there are several other papers on the topic, all of which are awesome and add to our jelly knowledge. I’ve compiled all the species from all these papers, and below is the full list of all the animals that have been reported to respond to each compound (of course, many species have yet to be tested). You can also read more about why these methods may work on some animals but not others in my first blog post.
Indomethacin:
Chrysaora pacifica
Chrysaora quinquecirrha
Chrysaora achlyos
Chrysaora fuscescens
Aurelia sp. N. Japan line
Aurelia sp. S. Japan line
Mastigias papua
Cephea cephea
Cassiopea sp.
Cotylorhiza tuberculata
Phyllorhiza punctata
Cyanea sp. Woods Hole
5M2M:
Cotylorhiza tuberculata
Chrysaora quinquecirrha
Aurelia sp. S. Japan line
Mastigias papua
Carybdea sp.
Cyanea sp. Woods Hole line
Thysanostoma thysanura
Rhopilema esculentum
Morbakka virulenta
Tripedalia binata
Tripedalia cystophora
What doesn’t it work on?
Coronate scyphozoans, so far, don’t respond. Though I’m curious hear what others find. Also one scypho was reported not to respond (likewise, I’d be curious to hear if this holds true for everyone). No hydrozoans tested so far respond to either compound, but that makes sense because hydroz don’t strobilate. Also the one anthozoan I tested didn’t do anything, because, you know, why not try?
Linuche unguiculata
Phacellophora camtschatica
Craspedacusta sowerbyi
Podocoryne carnea
Aequorea coerulescens
Aequorea macrodactyla
Aequorea victoria
Eutonina indicans
Tima formosa
Laodicea undulata
Melicertum octocostatum
Bougainvillia bitentaculata
Leuckartiara octona
Neoturris breviconis
Rathkea octopunctata
Sarsia tubulosa
Nematostella vectensis (I mean, it probably wouldn’t…)
What’s the recipe again?
(note the difference in volume between the two drugs)
For indomethacin:
“We prepared a 50 mM indomethacin stock solution by adding 18 mg of powdered indomethacin (Sigma-Aldrich, I7378-25G) to 1 mL DMSO. We added indomethacin stock solution to animal media at a ratio of 1 μl stock to 1 ml media for a final concentration of 50 μM. Indomethacin rapidly precipitates in water, so we stirred each treatment to facilitate rapid dissolving/resuspension of indomethacin.”
For 5M2M:
“We prepared stock solutions of 5-methoxy-2-methylindole (Sigma-Aldrich, M15451-5G) by adding 16 mg 5-methoxy-2-methylindole to 2 mL 100% ethanol, for a final concentration of 50 mM. We prepared working dilutions by adding 1 μL for every 1 mL medium, for a final concentration of 50 μM. We conducted dilutions of this final 50 μM to obtain 20 μM and 5 μM solutions.”
Any final thoughts?
As with anything in science, this is no where near the final method. So please keep me and others posted in the comment section below if you find something that works (or doesn’t!) And again, if you have any questions, I’ll be more than happy to respond (and will be sure to post any email questions/answers anonymously in the comments). Happy jelly growing!
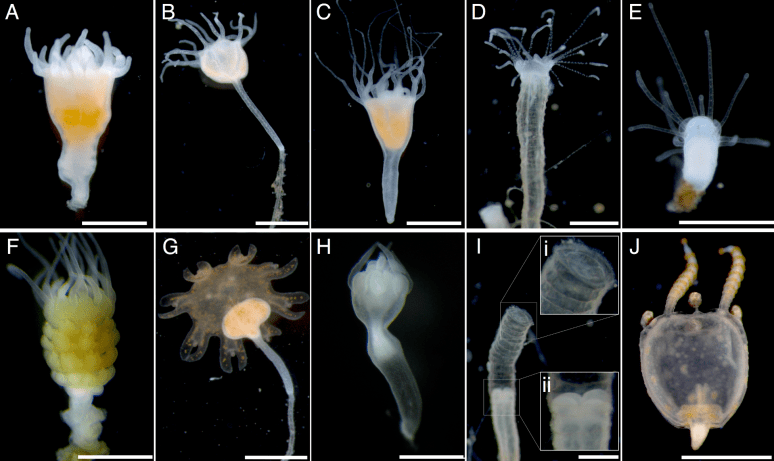
Fig 3. From my paper, showing control polyps on the top, and indomethacin-induced polyps/strobilae/medusae on the bottom.

On the topic of jellyfish in captivity… I had a few hitch hikers show up in my tank. They don’t get much bigger than the size of the tip of a ball point pen. About the size of copepods actually. I like them, they do seem to sting. However I am on the fence about keeping them. I had a peppermint shrimp that took out most of the population. How do I take care of them if I decide to keep them? How long is their life cycle? I have video I can send for identification upon request.
Hmmm yes videos or pics would be helpful!